Sumitomo Heavy Industries, Ltd. (Head office: Shinagawa-ku, Tokyo; President: Shinji Shimomura; hereinafter “Sumitomo Heavy Industries”) and STELLA PHARMA CORPORATION (Head office: Chuo-ku, Osaka; President: Koki Uehara; hereinafter “Stella Pharma”) have respectively entered into a sales contract for BNCT System, “NeuCure®“as well as “NeuCure® Dose Engine”, the BNCT dose calculation program and master service agreement for a boron drug for BNCT, “Steboronine®”, with China Biotech Services Holdings Limited, a company listed on The Stock Exchange of Hong Kong Limited, (Stock Code: 8037) and its subsidiary, Pengbo (Hainan) Medical Technology Co., Ltd. (hereinafter “Pengbo Co., Ltd.”), regarding the introduction of BNCT in Hainan Boao Lecheng International Medical Tourism Pilot Zone in China (hereinafter “Hainan Medical Tourism Pilot Zone”).
The Hainan Medical Tourism Pilot Zone is a special deregulated zone aimed at promoting medical tourism, with priority given to import of medical equipment and drugs.
A distinctive feature of the Hainan Medical Tourism Pilot Zone is that, similar to Japan, the use of any medical equipment or drugs in China require prior testing via clinical trials before applications can be filed. However, unlike the normal process of obtaining approval from the regulatory authorities, this system will grant import licenses for medical equipment and drugs that have already been approved in other developed countries on the premise that they are clinically urgently needed at designated medical institutions, even if they have not yet been approved in China. This therefore allows the medical institutions not to conduct clinical trials and to use such medical equipment and drugs for clinical treatment.
This time, the only globally approved BNCT, combination of NeuCure® and Steboronine® will be introduced in China through this system.
Pengbo Co., Ltd. will obtain a Medical Institution Practicing License and establish a designated medical institution (hereinafter “BNCT center”) in the Hainan Medical Tourism Pilot Zone. The land for the planned construction site has already been acquired, and the provision of BNCT is scheduled to start from FY2025 after the construction of the BNCT center and the installation of the BNCT system, “NeuCure®”, are completed.
The disease indicated for BNCT is “locally unresectable recurrent or unresectable advanced head and neck cancer” (hereinafter, “local head and neck cancer”), which is already approved in Japan. The total number of head and neck cancer patients in China is estimated to be around 140,000 per year. We will continue to develop BNCT as a new cancer treatment option for overseas patients who are waiting for it to be indicated as well.
If a new indication for BNCT is approved in Japan in the future, the treatment will also be made available in the Hainan Medical Tourism Pilot Zone similar to local head and neck cancer. In addition, the real world data obtained at the BNCT center in the Hainan Medical Tourism Pilot Zone can be used to apply for approval in China if the management, research, analysis, and evaluation are conducted in accordance with relevant regulations. This will help us to promote BNCT in the Hainan Medical Tourism Pilot Zone and use it as a foothold to expand BNCT into China.


Signing ceremony

【BNCT (Boron Neutron Capture Therapy)】
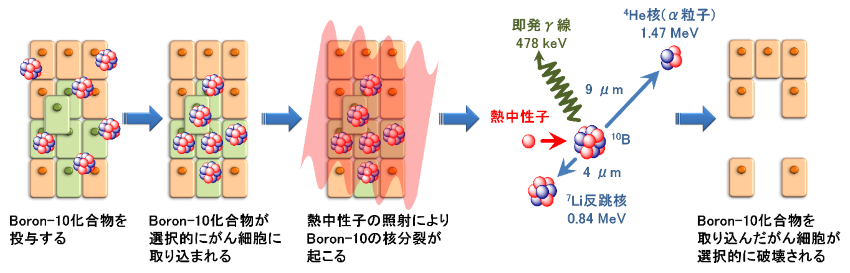
Boron-neutron capture therapy (BNCT) is a type of radiotherapy where the boron-containing drug for BNCT (Boron-10) is administered to cancer patients, which causes the selective uptake of boron (Boron-10) in cancer cells. A low-energy neutron beam is then irradiated on the cancerous area from outside the body. When the neutrons collide with the boron (Boron-10) in the cancer cells, nuclear fission occurs. This in turn generates alpha particles (helium nuclei) and lithium recoil nuclei (lithium nuclei) that damages cancer cells. These charged particles have a respective range of only about 9 µm and 4 µm in the body, which is approximately the size of a single cell. In theory, these characteristics allow us to selectively destroy cancer cells that have taken up boron (Boron-10) at the cellular level, with little to no damage to the surrounding normal cells.


【Overview of companies】